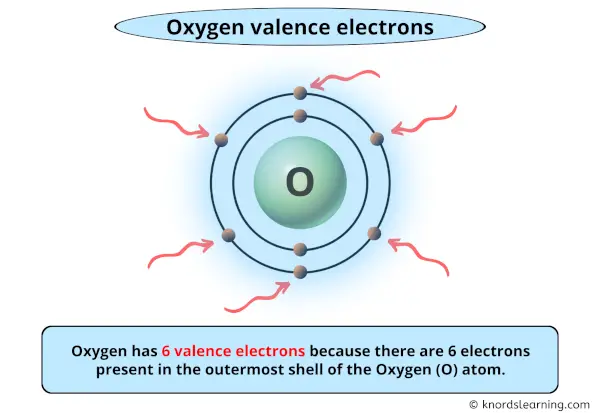 
Water contains nearly 89% of combined oxygen.It is a gas at room temperature, and about 1/4 of the atmosphere air by weight consists of oxygen.Oxygen comprises about 50% of the earth’s crust.It has two oxygen atoms in each of its molecules. At room temperature, oxygen is a non-metal element that exists as a gas.Since neither atom pulls harder, it’s a non-polar covalent bond. Equal electronegativity means that there are not any partial charges for each element. In the Oxygen molecule both oxygen atoms equally share the 4 electrons that make up the double bond between them. In the O2 molecule, both oxygen atoms have equal electronegativity and both atoms share equal ratios of bonded shared electrons and the overall molecule turns out to be nonpolar in nature. Oxygen is a diatomic molecule with linear molecular geometry and bond angles of 180 degrees. Therefore, two oxygen atoms are linked together through a double bond (one sigma and one pi bond).The remaining half-filled 2p orbitals undergo sidewise overlapping to form pi bonds.In the formation of the O2 molecule, one of the two half-filled 2p orbitals of each oxygen atom overlaps mutually along the internuclear axis to form a sigma bond.There are two half-filled 2p orbitals in the valence shell.The electronic configuration of the O2 atom (Z = 8) is 1s 22s 22p 4.The molar mass of oxygen is 15.9994 g/mol. 
Important Points- Oxygen Valence Electrons However, burning occurs only when the mixture of fuel and oxygen is hot enough. Burning requires the presence of oxygen.Respiration, the process of transferring energy from glucose to cells, requires oxygen.Its molecule contains two oxygen atoms.Oxygen is a non-metal element that is a gas at room temperature.Important Points- Oxygen Valence Electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
